-

Innovation, aesthetics, and safety at your disposal
We are leader in the production of professional devices for medical and aesthetic treatments.
-

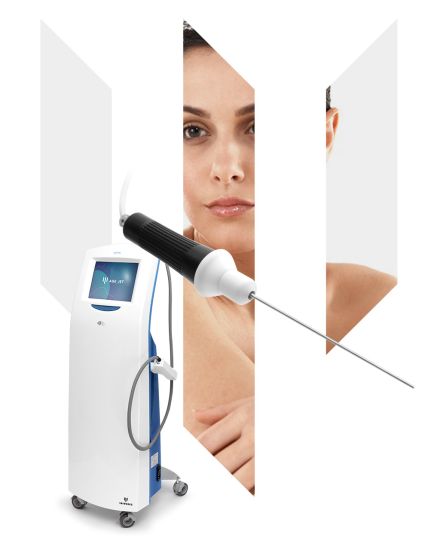
AGE JET
Plasma technology is one of the most effective mini-invasive treatments for rejuvenating and tightening skin.
-

OMNIKA
Two technologies that can be used simultaneously for body contouring: Cavitation and Radio Frequency.
-

DEROS
Equipped with two sophisticated handpieces for firming, vascularising, draining and hydrating compromised tissue.
-

MEDICAL RF
A versatile and effective alternative to surgical skin rejuvenation methods for gynaecological conditions.
-

Innovation, aesthetics, and safety at your disposal
We are leader in the production of professional devices for medical and aesthetic treatments.
-

AGE JET
Plasma technology is one of the most effective mini-invasive treatments for rejuvenating and tightening skin.
-

ONMIKA
Two technologies that can be used simultaneously for body contouring: Cavitation and Radio Frequency.
-

DEROS
Equipped with two sophisticated handpieces for firming, vascularising, draining and hydrating compromised tissue.
-

MEDICAL RF
A versatile and effective alternative to surgical skin rejuvenation methods for gynaecological conditions.
Technology at the heart of beauty.
From Central Italy to key international markets, Triworks focuses on the production and distribution of specialised medical technologies for professional medical and cosmetic treatments.
20 years in business alongside health personnel, researchers, biomedical engineers and experts in advanced cosmetic treatments have provided Triworks with the expertise that is strategic for continually developing equipment for the sector on the basis of specific, shared values.
Research
Science, innovation and foresight: aspects which are essential in the approach Triworks has taken since 2012
Design
Functionality, comfort, performance: crucial factors when designing any Triworks device
Safety
Precision, diligence, quality: key elements in all engineering, production and support phases
Optimisation and innovation.
Triworks devices result our willingness to listen to professionals all over the world.
Machines and equipment for professional cosmetic surgery and aesthetic treatments developed with the aim of improving performance, patient comfort, timing and mode of use.
Made in Italy,
used throughout the world.
Triworks was established in Rome and has grown in Molise. Rooted in the area, the company continues to develop the talent and skills of engineers, researchers, healthcare personnel and specialists in Central Italy.
High Italian standards in research and production are combined with a global presence through a structured network of distributors, and by closely engaging with cosmetic healthcare practitioners, dermatologists, plastic surgeons, specialist facilities and medical and cosmetic research partners throughout the world.

CONTACT
All-round support.
From pre-sales to after-sales, Triworks accompanies professionals in the choice and installation of equipment, providing the appropriate training with specialised engineers and a prompt and ongoing support and maintenance service.








